We use cookies to operate this website and to improve its usability. To see what cookies we use and set your own preferences, please refer to ourPrivacy & Cookies pagePlease note that by using this site you are consenting to the use of cookies.。Accept!
360° Integrated Skincare Solutions

Customized Ingredient Development
In-depth research and years of practical experience
To assist brands in developing exclusive and high active ingredient
- INCI application.
- Patent application.
- COSMOS application.
- HALAL application.
- Advanced Extraction Technology Platform
for optimal and efficient extraction. - Establish standard specifications to
ensure the stability of ingredient. - Ensure the efficacy and safety of
ingredient along with
Efficacy&Safety Validation Platform. - Qualitative and quantitative analysis.

Efficacy Evaluation
Scientific validation through our BSL2 laboratories and collaboration with medical institutions for clinical trials
- Anti-Acne Evaluation
- Anti-Oxidant Evaluation
- Collagen Synthesis Activity
- Whitening Efficacy Evaluation
- Skin Moisture Evaluation
- Anti-inflammatory Evaluation
- Skin Cellular Repair Effectiveness
Evaluation - Anti-aging Activity Evaluation
(Anti-photoaging/ Anti-wrinkle) - The Content of Polysaccharide/
Total Polyphenol / Flavonoids Assay - Cutaneous Absorption/ Penetration Property Test
- Skin Barrier Function Test

Safety Assessment
Established alternative animal test, international OECD Test Guidelines, to evaluate the safety of ingredient.
Cooperated with International clinical laboratories to conduct human clinical tests.
- Patch Test
- Ames Test
- Phototoxicity Test
- Photosensitivity Test
- Cell Viability Assay
- Repeat Insult Patch Test, RIPT
- In Vitro Skin Irritation Test(OECD 439)
- In Vitro Eye Irritation Test (OECD 492)
- In Chemico Skin Sensitisation Test
(OECD 442C)
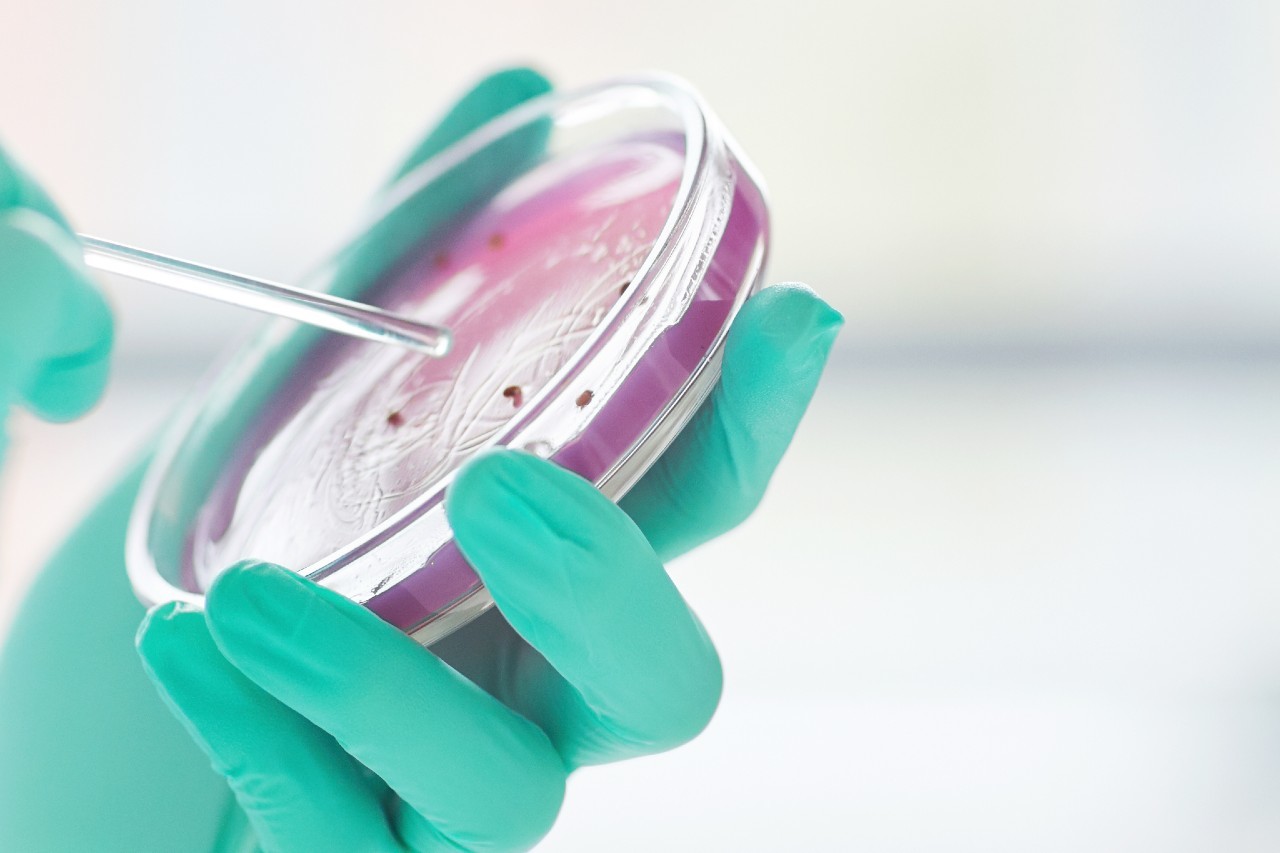
Microbiology Testing
Established BSL2 Microbiology Lab
Assist customers in solving a variety of microbiological problems
- Antimicrobial Test
(five main types of pathogens/P.acnes/M. furfur) - Microbiological Test
(A. niger/ S. aureus/ E. coli/ P. aeruginosa/ C.albicans) - Preservative Development conform to ISO 11930
(MIC Test/ Zone of inhibition/
Preservative Efficacy Test/
Microbial Test) - Skin Probiotics Development

INCI Name Application
Facilitate the global distribution of unique ingredients and increase consumer awareness
- Organize data sheets and
technical information for
processing the applications. - Customized ingredient development
including INCI applications.

Compliance Service
Regulate to ensure the highest level of consumer safety.
Assist customers become a trusted and valued cosmetic brand.
- EU PIF service
- TW PIF service
- Imported Cosmetics registration
and filing in China - ASEAN Cosmetics Registration
Service - TFDA Cosmetic Compliance Service(PIF filing/Registration of
Cosmeceutical)

Private Label
Develop regulatory-compliant, cost-effective and high-performance products.
- Formulation Type:Liquid/Emulsion/
Cream/Oil - Category:Skin Care/Body Care/
Hair or Scalp Care - Customized product proposals based on
brand needs and market trends - Special Treatment Product Development
and Design- After laser treatment products
- EU Cosmos verified products
- Preservative-free products
- Gentle products for sensitive skin
- Hair and scalp care
- Feminine care and anti-dandruff
products

